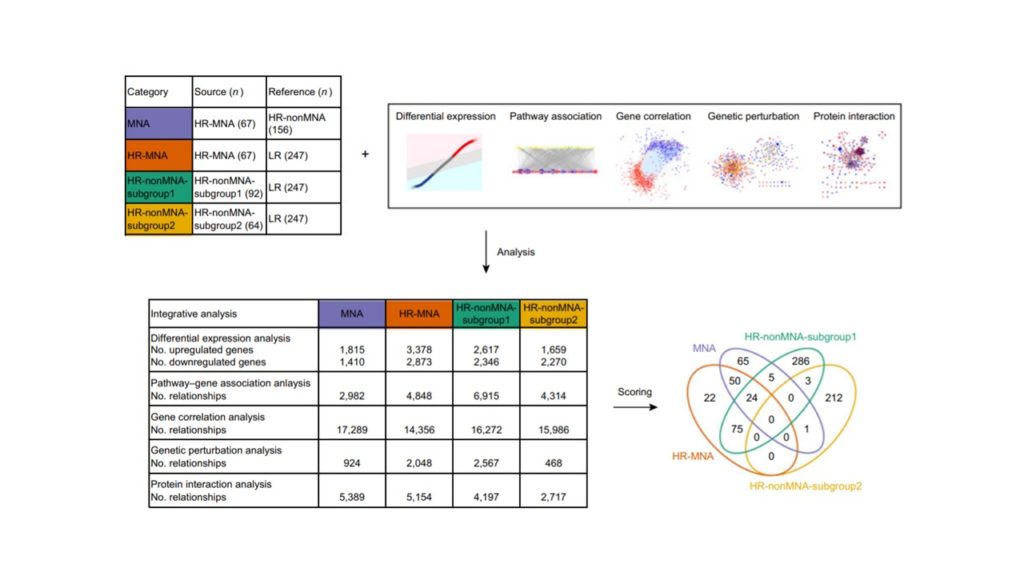
研究主題:Therapeutic Targeting of Non-oncogene Dependencies in High-risk Neuroblastoma神經母細胞瘤是一種源於周圍交感神經系統的惡性腫瘤,發生於兒童早期。而MYCN致癌基因的擴增已被證明是驅動神經母細胞瘤的發病機制之一,並與高危險群的患者相關。然而,由於缺乏突變和分子異質性,使得標靶治療的發展受到限制。為了找到新穎且有效的治療方法,本研究從GEO資料集中對近千名原發性神經母細胞瘤患者進行綜合分析,並預測針對高風險神經母細胞瘤非癌基因依賴性的潛在藥物。此外,在老鼠體內驗證了耐克螺(一種FDA批准治療絛蟲感染的驅蟲藥物)的抗神經母細胞瘤活性,並透過蛋白質體學研究其機制。該研究證明利用原發神經母細胞瘤的轉錄圖譜和小分子進行藥物開發的可能性,並為耐克螺治療神經母細胞瘤的臨床療效提供了機會。Neuroblastoma is a malignant tumor arising in the peripheral sympathetic nervous system. It typically occurs in early childhood. It has been shown that genomic amplification of MYCN oncogene driving neuroblastoma pathogenesis. And it is related to stratify patients into neuroblastoma risk groups. However, the development of targeted therapies is limited due to the paucity of mutations and molecular heterogeneity. To find new therapeutic approaches and effective treatments, this research performs integrative analysis of nearly a thousand patients with primary neuroblastoma tumors obtained from multiple Gene Expression Omnibus (GEO) datasets and identify potential drugs that target non-oncogene dependencies in high-risk neuroblastoma. Furthermore, the therapeutic efficacy of niclosamide, an anthelmintic drug approved by the FDA to treat tapeworm infections, is also demonstrated in vivo and investigated the mechanism through proteomics. The study exhibits the feasibility of using transcriptional profiling of primary neuroblastomas and small-molecule treatments for drug discovery and provides an opportunity for the clinical efficacy of niclosamide in the treatment of high-risk neuroblastoma.
Graduate Institute of
Biomedical Electronics and Bioinformatics
National Taiwan University
國立臺灣大學生醫電子與資訊學研究所